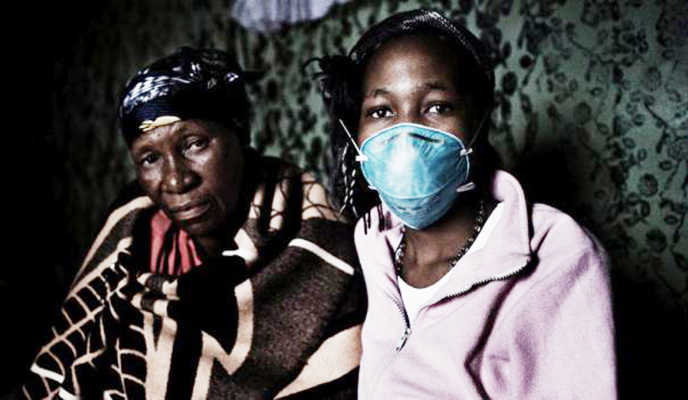
Humanitarian organisations and health activists have expressed concern at the exorbitant cost of the new life-saving tuberculosis (TB) drug, Delamanid, which is considered the most effective in fighting multi-drug resistant TB which is wreaking havoc in countries such as Zimbabwe.
By Phyllis Mbanje
Japanese pharmaceutical company Otsuka said it would make Delamanid available to some developing countries at a price of $1 700 per treatment course.
Delamanid is one of only two new drugs to treat the deadliest strains of tuberculosis that are resistant to many of the other drugs used to treat TB, including multidrug-resistant and extensively drug-resistant TB.
However, international medical humanitarian organisation Médecins Sans Frontières (MSF) has expressed concern at the high price of the drug which is unaffordable for countries like Zimbabwe.
Delamanid needs to be taken with several other drugs to effectively treat drug-resistant TB.
“Countries should start scaling up treatment for more people with drug-resistant TB using the most effective drugs available, but Delamanid is neither affordable nor available in most countries today,” Grania Brigden, TB adviser for MSF’s Access Campaign said.
- Chamisa under fire over US$120K donation
- Mavhunga puts DeMbare into Chibuku quarterfinals
- Pension funds bet on Cabora Bassa oilfields
- Councils defy govt fire tender directive
Keep Reading
She emphasised that the price for Delamanid needed to come down to an affordable level and that Otsuka should also register it quickly in all countries where the drug has been tested in clinical trials, as well as in countries with the highest burdens of drug-resistant TB.
“If people can’t access Delamanid, this promising new drug will be effectively worthless,” Brigden said.
Countries that are eligible for funding from the Global Fund to Fight Aids, Tuberculosis and Malaria (GFATM) will be able to purchase Delamanid for $1 700 per treatment course through the Global Drug Facility, a United Nations-based procurement mechanism for TB drugs, as long as the drug is registered for use in their country or the necessary import waivers have been put in place.
But unfortunately for countries like Zimbabwe, Otsuka has registered Delamanid in Germany, Japan, South Korea, and the United Kingdom, none of which have a high burden of drug-resistant TB.
“Otsuka should prioritise expanding access for people whose lives could be saved by Delamanid,” Brigden said. “Every effort should be made to ensure as many people as possible can benefit from this promising new treatment, but that’s unfortunately not what we’re seeing today.”
Two thirds of the nearly half a million people who contract drug-resistant TB each year could benefit from Delamanid but, in the two years since the drug was approved, only 180 people have received this new treatment.












